An early version of the twin-turbine, rotary total artificial heart was implanted in a calf to test durability and function.
Extending Continuous Flow to Total Heart Replacement
Early efforts to answer that question involved replacing the excised heart with two independent continuous-flow pumps—one for the systemic circulation and one for the pulmonary circulation.
Over roughly 8.5 years, Dr. Cohn, Dr. Frazier, and a team of collaborators implanted such systems in 68 large-animal models. These experiments were technically demanding and often unforgiving. Only 30 animals survived longer than 1 week, and infections were common.
Yet the experiments yielded critical insights. Contrary to long-standing concern, balancing right- and left-sided flows was not the dominant challenge. Continuous-flow pumps exhibit intrinsic automaticity. As inflow pressure rises, flow increases without a change in revolutions per minute, mimicking a Starling-like response. Thus, many animals maintained stable hemodynamics without constant adjustment.
The major Achilles’ heel was ingested thrombus on the right side. Small venous clots—subclinical pulmonary emboli that mammals generate routinely—could lodge in tight-clearance turbines and abruptly halt flow. That observation drove the next design leap: reducing complexity even further.
Single Rotor, Levitated in Space
The pivotal shift came with the arrival of Dr. Timms from Australia. Motivated by his father’s death from heart failure, Dr. Timms had independently conceived of a titanium-constructed total artificial heart built around a single, double-sided impeller capable of pumping blood to both circulations. The rotor would be magnetically levitated—held in space by electromagnets responding to high-frequency sensor feedback—eliminating physical contact, bearings, and mechanical wear. In addition, the right-sided impeller was designed to allow thrombus to pass without affecting right pump function.
“It’s like science fiction,” Dr. Cohn said. “There’s no mechanical wear. The rotor never touches anything. There’s no reason it shouldn’t last indefinitely.”
When Drs. Cohn and Frazier heard the concept articulated, they recognized its potential, and Dr. Timms, who is now the founder and chief technology officer of BiVACOR, arrived in Houston. They committed fully to the new architecture.
“Big projects have humble beginnings,” shared Dr. Cohn, BiVACOR chief medical officer. “And when you meet someone brilliant who is working on something that excites you, don’t let them leave your side.”
What followed was an intense period of digital design and rapid prototyping. Using computer modeling and 3D printing, the team produced dozens of impeller and volute geometries, testing each variant on the bench. Subtle changes in shape produced dramatic differences in flow efficiency, shear stress, and thrombogenic potential. Computational fluid dynamics informed the process, but bench testing with real blood remained the definitive arbiter. Models often failed to predict critical real-world behaviors.
Dr. Daniel Timms invented the world's first durable total artificial heart, which is currently being tested for trials in patients.
Once geometry was optimized, the team transitioned to metal prototypes using additive manufacturing. Titanium pumps emerged layer by layer from laser sintering machines, then were polished and implanted in animals. Outcomes improved steadily.
After approximately 40 successful implants, regulators required five consecutive animals to survive 30 days. The team met that benchmark, clearing the way for first-in-human experience.
According to BiVACOR, the size of the total artificial heart is suitable for most men and women (body surface area >1.4 m2). Despite its small size, the total artificial heart can provide enough cardiac output for an adult male who is exercising. Using magnetic levitation technology, the same principle used in high-speed trains, the product features a unique pump design with a single moving part. A magnetically suspended dual-sided rotor with left and right vanes positioned within two separate pump chambers forms a double-sided centrifugal impeller that moves blood from the respective pump chambers to the pulmonary and systemic circulations.5
The total artificial heart has no valves or flexing ventricle chambers, with magnetic levitation and motor control making pulsatile outflow possible by rapidly modulating the rotor’s rotational speed once per second. The noncontact suspension of the rotor is designed to eliminate the potential for mechanical wear and provide large blood gaps that minimize blood trauma, offering a durable, reliable, and biocompatible heart replacement. A small external controller, combined with a rechargeable battery system, supports untethered operation from an AC power source to enhance patient mobility and freedom of movement.6
Early Clinical Experience—and Hard Lessons
Under an FDA early feasibility framework, four US centers with deep artificial heart and transplant expertise were activated. The cohort of five patients all suffered from severe biventricular failure and faced imminent death. Several were supported preoperatively with temporary devices and were deteriorating from right-sided failure.
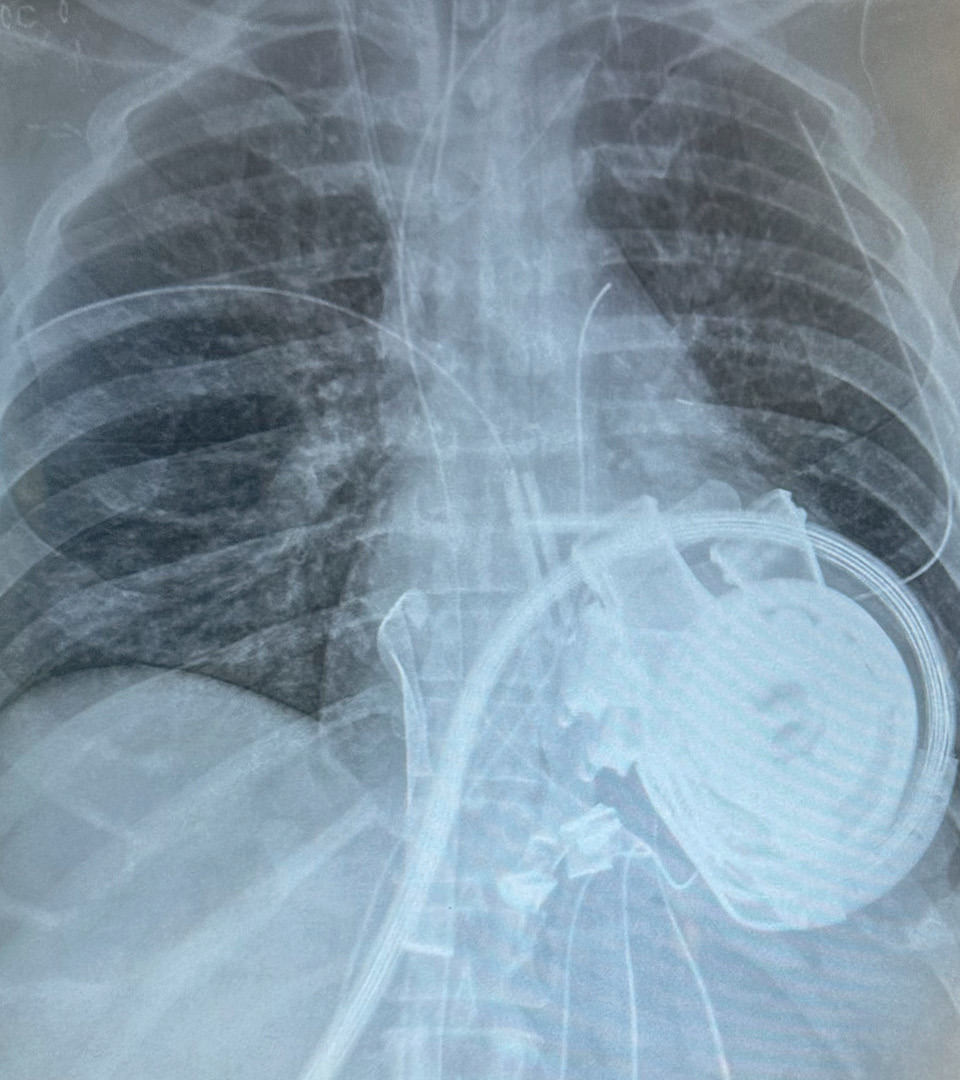
A postoperative chest x-ray shows an implanted BiVACOR total artificial heart and its driveline connections.
The first candidate in the study was a 58-year-old man who had suffered from end-stage heart failure. Surgeons at The Texas Heart Institute, including Drs. Cohn and Frazier, successfully implanted the total artificial heart in the patient. The device helped him maintain normal vital signs and organ function for 8 days—until he received a lifesaving heart transplant. The device was tested in four additional patients as well.7
Post-implantation, recovery was striking. Patients were mobilized early, walking in hallways within days. One patient walked nearly 2 miles per day. Cardiac output increased autonomously with exertion, without manual pump adjustments. By modulating pump speed, clinicians were able to generate a palpable arterial pulse—sometimes referred to as “digital pulse”—allowing conventional blood pressure cuffs and pulse oximetry to function normally.
“One patient told us, ‘I don’t have a heart, literally, and I have not felt this good in I don’t know how long,’” Dr. Cohn said.
Because of trial design, US patients remained in the ICU and were rapidly relisted for transplant, resulting in relatively short support durations. While the study was originally designed to include five patients, an additional 15 patients were added in late 2024. Those patients are expected to receive their devices in the months ahead.
In mid-2025, the titanium total artificial heart received the FDA’s Breakthrough Device designation—a formal identification that a device in development should be expedited for patient access.
“This is more than a regulatory milestone. It’s a validation of a concept we’ve spent decades proving that a fully implantable, total artificial heart isn’t just possible, it’s necessary,” Dr. Timms said in a statement.8 “The early results from our clinical trial show that we can give them a second chance, without the compromises of older technologies. The Breakthrough Device designation puts us on a faster track to deliver exactly that.”
Parallel experience in Australia allowed discharge home; one patient lived more than 100 days with the device before transplantation.
Not all outcomes were favorable, though. Two later patients in Australia died from intracranial hemorrhage several weeks after implantation. Independent review determined the events were not device-related, implicating blood pressure and anticoagulation management, but the losses were devastating nonetheless. For Dr. Cohn, they underscored that novel devices demand not only engineering excellence, but also rigorous clinical protocols and constant refinement.
The surgical team at The Texas Heart Institute works with Dr. William Cohn to prepare for a total artificial heart implant.
Toward Destination Therapy
Despite setbacks, momentum continues. Power requirements for the device are dramatically lower than prior total artificial hearts—on the order of one watt per liter per minute of flow—making transcutaneous energy transfer via inductive coupling increasingly feasible. Ongoing miniaturization already has moved most computing capability into the device itself, with future generations aiming to eliminate external drivelines entirely.
The ultimate ambition is not simply to bridge patients to transplant, but to offer a viable alternative. For the hundreds of thousands of patients with end-stage heart failure who will never receive a donor heart, a small, durable, energy-efficient and blood-friendly artificial heart could redefine the standard of care.
“Our goal is not to be a bridge to transplant. We think this device could possibly be better than heart transplant and become the gold standard,” said Dr. Cohn.
The journey also has carried symbolic weight for Dr. Cohn. Decades after the falling-out between Dr. DeBakey and Dr. Cooley, both men ultimately stood together at the bedside of an experimental implant, reconciled by the very technology that once divided them—a moment Dr. Cohn described as a reminder that high-stakes innovation can shape not only technology, but institutions and relationships as well, while also a signal that the long pursuit of a total artificial heart is finally approaching its most consequential chapter.
“With breakthrough status in hand,” said Dr. Cohn, “we’re entering the next phase with the wind at our backs and real momentum to bring this to more patients.”
Dr. Cohn delivered the I. S. Ravdin Lecture in the Basic and Surgical Sciences, “The Past, Present, and Future of the Total Artificial Heart: A Very Houston-Centric Story,” at the 2025 ACS Clinical Congress in Chicago, Illinois. This presentation was featured in episode 68 of The House of Surgery® podcast.