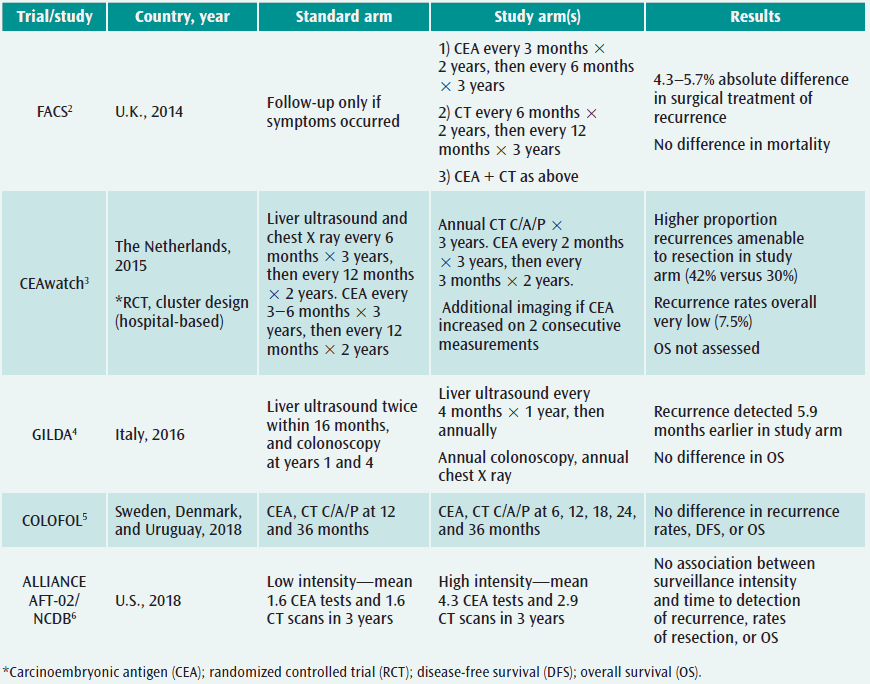
FACS trial
The first of these is the Follow-up after Colorectal Surgery (FACS) trial, a randomized controlled trial of patients treated at 39 National Health Service hospitals in the U.K.2 Patients treated with curative resection for primary colorectal cancer were randomized to one of four groups consisting of follow-up with CEA levels, CT scans, CEA levels and CT scans in combination, or testing only if symptoms occurred. Rates of curative surgical treatment of recurrence ranged from 6.6–8.0 percent in the testing groups versus 2.3 percent in the minimum follow-up group. However, the absolute difference in curative resection of recurrence measured only 5 percent, and the number of deaths did not differ between the more intensive monitoring cohorts and the minimum follow-up cohort.
CEAwatch trial
The CEAwatch trial used a unique study design, in which clusters of hospitals sequentially transitioned from a usual follow-up regimen to an intensified follow-up regimen for patients with resected colorectal cancer.3 Standard follow-up consisted of an outpatient clinic visit with liver ultrasound and chest X-ray every six months and CEA every three to six months in the first three years. The intervention protocol included bimonthly CEA measurements and annual CT imaging of the chest and abdomen in the first three years, with additional imaging ordered if CEA increased more than 20 percent over two consecutive measurements. A higher proportion of the total number of recurrences were detected among patients followed at hospitals using the more intensified surveillance schedule (57 percent versus 43 percent), and a greater proportion of recurrences in the intensified surveillance group were definitively treated with curative intent (42 percent versus 30 percent). However, the generalizability of these results is limited, as the overall recurrence rate in the study (7.5 percent) is much lower than most published recurrence rates, which typically average 20 percent. Furthermore, this study was not designed or powered to assess overall survival.
GILDA trial
In the Italian GILDA trial, 1,228 patients were randomized to either intensive surveillance with annual colonoscopy and ultrasound and chest imaging every six months or minimum follow-up with two ultrasounds within 16 months and colonoscopy at years one and four.4 In this study, recurrence was detected an average of 5.9 months earlier (95 percent confidence interval [CI] 2.7–9.1) among patients followed with more frequent testing. However, this small difference in disease-free survival was statistically insignificant, nor was any difference detected in overall survival.
COLOFOL trial
The highly anticipated, randomized multicenter COLOFOL trial reported similar results, which were published this May in the Journal of the American Medical Association.5 In this study, 2,509 patients were randomized to follow-up testing with CT scans of the chest and abdomen and CEA levels every six months for three years or the same tests at only 12 and 36 months after initial resection. Recurrence rates were equivalent in the high versus low frequency testing groups (21.6 percent versus 19.4 percent), as was disease-specific and overall mortality (10.6 percent versus 11.4 percent and 13 percent versus 14.1 percent, respectively).
Alliance and CoC collaboration
A collaborative effort of the Alliance Surveillance Optimization Working Group (AFT-02) and the Commission on Cancer gave rise to a recently published large, national study of surveillance testing intensity within the National Cancer Database and demonstrated similar results.6 In this study, cancer registrars collected primary data on colorectal cancer recurrence, a data element not routinely available in U.S. cancer registry datasets. Among 8,529 patients treated at CoC hospitals, the median time to detection of recurrence among patients treated at hospitals providing high-intensity CT surveillance was 15.1 months and high-intensity CEA surveillance 15.9 months, compared with 16 months for patients treated at low-intensity CT surveillance and 15.3 months for low-intensity CEA surveillance facilities. Again, no significant differences in rates of resection for cancer recurrence or overall survival were identified.
Impact and future directions
These studies present collective evidence that more frequent or intensive surveillance testing may not result in earlier detection of recurrence that leads to a clinically meaningful benefit for the patient. Informing a patient of cancer recurrence earlier, without ultimately improving the long-term outcome, could even be considered detrimental in some cases. Furthermore, more frequent testing has known disadvantages. In addition to increasing health care costs, patients (and their caregivers) may miss more days at work; experience more anxiety around the time of testing; be subject to false-positive results requiring additional unnecessary testing; and be exposed to higher cumulative radiation dosage over time.
Inarguably, surveillance testing has its benefits, but the optimal frequency of testing remains in question. Given recent evidence, it may be time to reevaluate U.S. consensus guidelines to more closely align with other guidelines around the world, such as those that the National Institute for Health and Care Excellence in the U.K. has issued, which advocate for CEA testing every six months and two CT scans during the first three years of survivorship.7 Further, individual risk-stratification to include a patient’s underlying health and performance status, as well as tumor elements associated with clinical risk of recurrence—such as American Joint Committee on Cancer Stage, tumor location, and molecular or genomic profile—may be warranted.
References
- Buie WD, Attard JP. Follow-up recommendations for colon cancer. Clin Colon Rectal Surg. 2005;18(3):232-243.
- Primrose JN, Perera R, Gray A, et al. Effect of 3 to 5 years of scheduled CEA and CT follow-up to detect recurrence of colorectal cancer: The FACS randomized clinical trial. JAMA. 2014;311(3):263-270.
- Verberne CJ, Zhan Z, van den Heuvel E, et al. Intensified follow-up in colorectal cancer patients using frequent Carcino-Embryonic Antigen (CEA) measurements and CEA-triggered imaging: Results of the randomized “CEAwatch” trial. Eur J Surg Oncol. 2015;41(9):1188-1196.
- Rosati G, Ambrosini G, Barni S, et al. A randomized trial of intensive versus minimal surveillance of patients with resected Dukes B2-C colorectal carcinoma. Ann Oncol. 2016;27(2):274-280.
- Wille-Jørgensen P, Syk I, Smedh K, et al. Effect of more vs less frequent follow-up testing on overall and colorectal cancer-specific mortality in patients with Stage II or III colorectal cancer: The COLOFOL randomized clinical trial. JAMA. 2018;319(20):2095-2103.
- Snyder RA, Hu CY, Cuddy A, et al. Association between intensity of post-treatment surveillance testing and detection of recurrence in patients with colorectal cancer. JAMA. 2018;319(20):2104-2115.
- National Institute for Health and Care Excellence. Colorectal cancer: Diagnosis and management. Clinical guideline (CG131). Available at: www.nice.org.uk/guidance/CG131. Accessed October 12, 2018.